
It is important to underline that a registered side effect does not necessarily mean the effects were triggered solely by the vaccine, or are connected to the vaccination in any way, but as they occurred following a vaccination they must be registered.
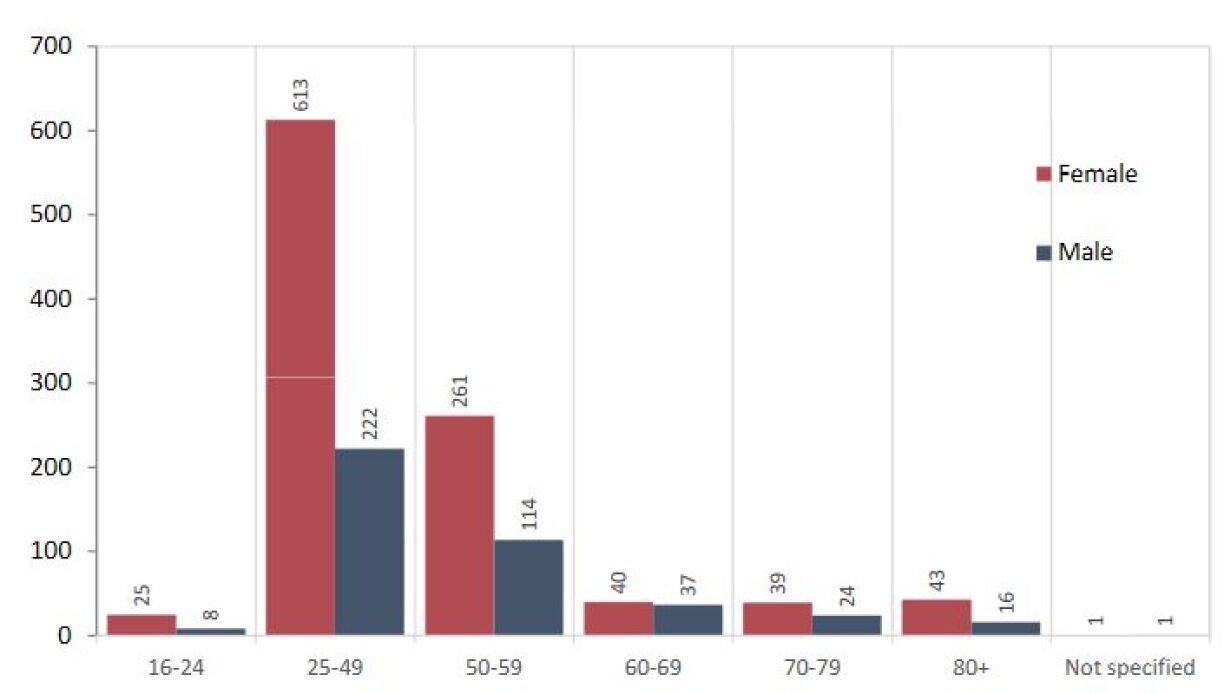
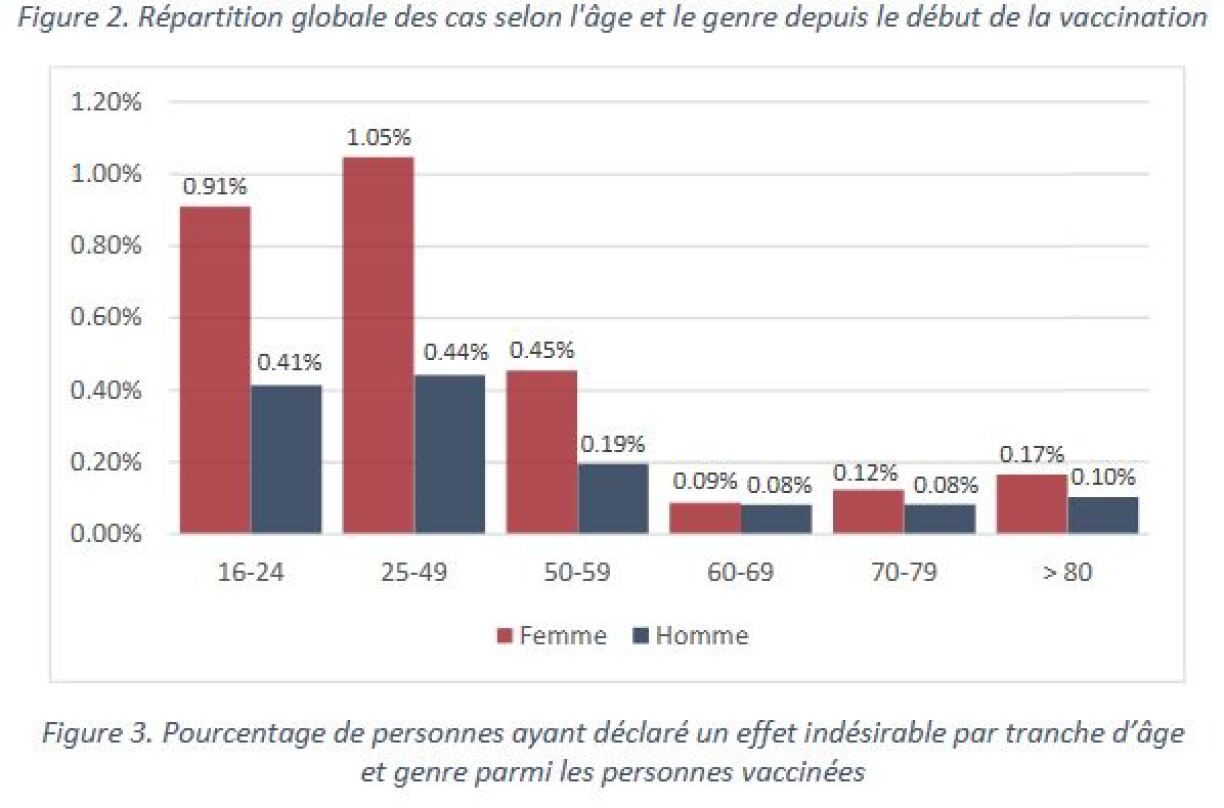
1,444 possible cases of side effects were registered since the start of the vaccination campaign, which corresponds to 0,34% of all administered doses. 1,022 cases affected women, and 422 affected men. The age categories 16-24 and 25-49 are most prevalent.
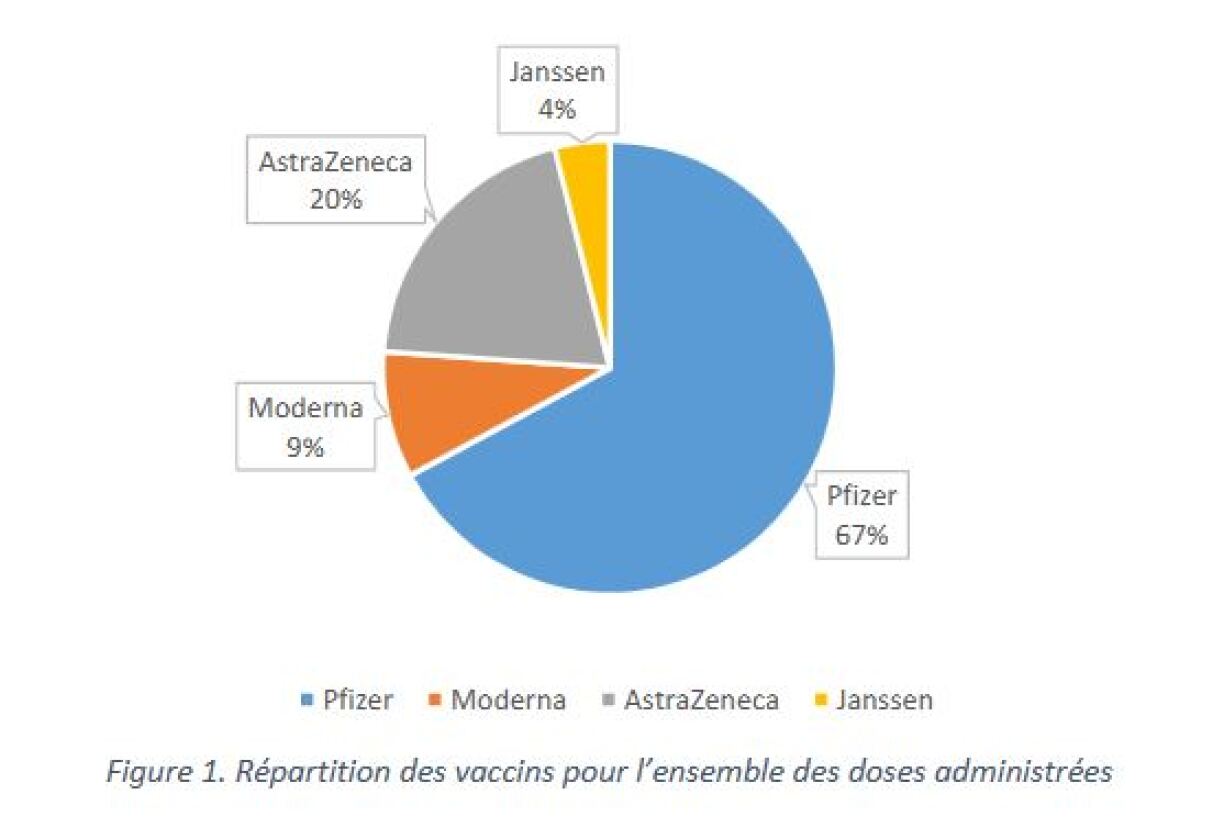
As of 11 June, Luxembourg has administered 423,148 vaccine doses in total - 284,080 doses of BioNTech/Pfizer, 31,117 doses of Moderna, 84,309 doses of AstraZeneca and 16,642 doses of Janssen (Johnson & Johnson). 263,375 people have been vaccinated at least once.
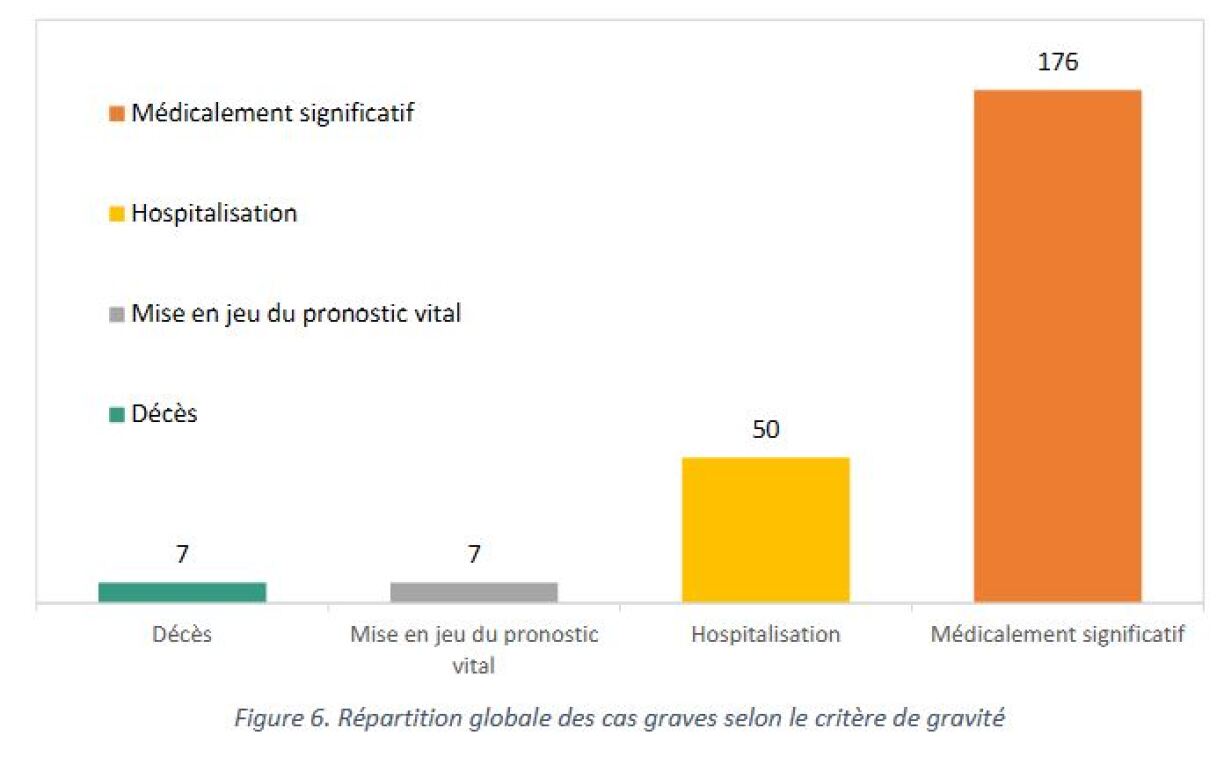
Out of the 1,444 cases, 1,204 cases were categorised as "not severe", and include mostly flu-like symptoms or digestion problems. 240 cases, or 17%, were categorised as "severe". 50 people ended up hospitalised, and there were 7 deaths. Again, this does not imply that their was a causal link between their death and receiving the vaccination.
Taking a look at the cases of side effects and the vaccines that were used in those instances, that of BioNTech/Pfizer comes first with 1,236 cases, but this is also the vaccine that has been administered the most. Side effects were registered in 4 cases out of every 1,000 jabs with BioNTech/Pfizer - 872 cases with women and 364 with men.
The Moderna vaccine, which represents 9% of all administered jabs, may have been responsible for 45 instances of side effects, translating to 1 in every 1,000 jabs. 157 cases of side effects were reported following AstraZeneca, and six cases after Janssen (Johnson & Johnson).
The report also notes that non-serious side effects have a 90-day window within which they are reported, which means the number may be higher. However, these effects tend to be in line with known side effects that signal a healthy immune system response to the vaccine.